 Sometimes there may be multiple compounds with that element on one side, so you'll need to use your best judgement and be prepared to go back and try the other options. The cesium atom has a radius of 265 pm and a Van der Waals radius. The number of electrons in each of Cesium's shells is 2, 8, 18, 18, 8, 1 and its electron configuration is Xe6s 1. Multiply coefficients for compounds to balance out each elementįor each element that is not equal, try to balance it by adding more of it to the side with less. Cesium (or Caesium) (atomic symbol: Ce, atomic number: 55) is a Block S, Group 1, Period 6 element with an atomic weight of 132.9054519. Count the number of each element on the left and right hand sidesĢ. Caesium gets its name from the Greek for heavenly blue. Caesium sulfate or cesium sulfate is the inorganic compound and salt with the formula Cs 2 SO 4. substitutue 1 for any solids/liquids, and Psubstance for gases. K c or Q ( Cs 2 S ) / ( Cs 2 S ) (assuming all reactants and products are aqueous. Equilibrium Constant & Reaction Quotient. What names and formulae of the two products of the reaction between caesium and water 2 Cs + 2 H2O &rarr. Cesium Sulfide Caesium Sulfide Cs2S Molar Mass Cs2S Oxidation Number. CESIUM SULFIDE CAS 1 WIKI information includes physical and chemical properties, USES, security data, NMR spectroscopy, computational chemical data. the amount of heat given off increases as you go from lithium to caesium. When using the inspection method (also known as the trial-and-error method), this principle is used to balance one element at a time until both sides are equal and the chemical equation is balanced. The chemical formula of caesium sulfide is Cs2S. This equation applies to any of these metals and water - just replace the X by. To be balanced, every element in CsCl + CaS = Cs2S + CaCl2 must have the same number of atoms on each side of the equation. Cesium sulfate 99. 430.The law of conservation of mass states that matter cannot be created or destroyed, which means there must be the same number atoms at the end of a chemical reaction as at the beginning. This problem has been solved Youll get a detailed solution from a subject matter expert that. Auerbach: 'Handbuch der anorganischen Chemie'. Use Lewis Dot Symbols to show the formation of Caesium sulfide.  
308, 705, 712 caesium chloride, 181 chloride, 308 double chlorides.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

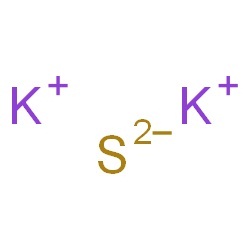
 RSS Feed
RSS Feed
